
For nearly 31,000 years, humans have documented cases of lower limb amputation. The Capua leg, discovered in a grave near Capua, Italy, is the earliest evidence of lower extremity prosthetics and dates to 300 BC. Yet humans had not ventured beyond this traditional socket prosthetic technology until recently.
Successful lower limb amputation may be initially defined by a stable, well-healed soft tissue envelope that can tolerate a socket prosthesis. But for many patients, amputation begins a lifelong battle with socket wear issues, painful residual limbs (including skin breakdown), phantom limb pain, and complaints of prosthetic heaviness or control issues. Many members of orthopaedic, vascular, and plastic surgery teams have seen these patients in clinic: transfemoral amputees who, despite receiving a technically sound surgery, are still unable to tolerate a socket prosthetic due to this laundry list of problems.
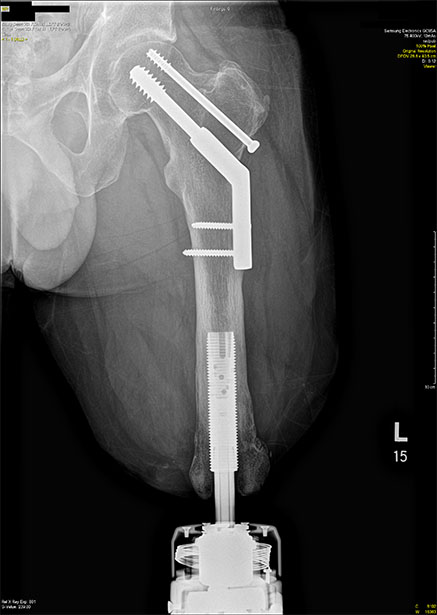
OPRA: Osseointegrated Prostheses for the Rehabilitation of Amputees
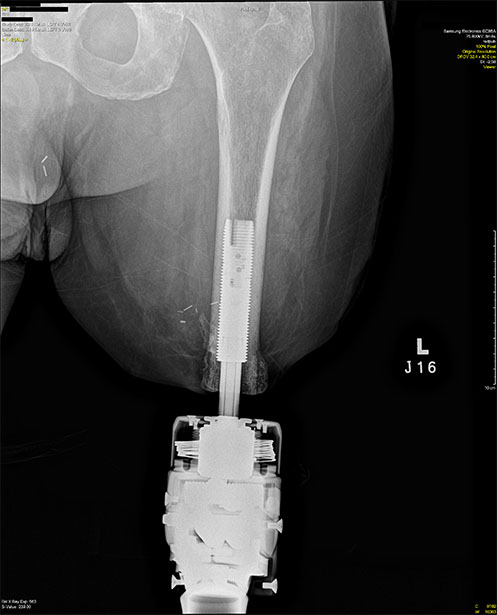
OPRA: Osseointegrated Prostheses for the Rehabilitation of Amputees



Many secondary surgical treatments are available for residual socket issues, including soft tissue revision and peripheral nerve surgery optimization. Targeted muscle reinnervation (TMR) and regenerative peripheral nerve interfaces (RPNIs) are often performed concurrently with amputations. TMR connects severed endings of the peripheral nerves at any level to motor branches of adjacent muscles, while RPNI wraps these endings with autologous muscle grafts. Regenerating axons now have a physiologic target, which can significantly decrease painful neuroma formation or phantom limb pain development. Patients directly benefit from decreased neuropathic pain post-amputation, possibly even experiencing functional benefits that encourage prosthetic control down the line.
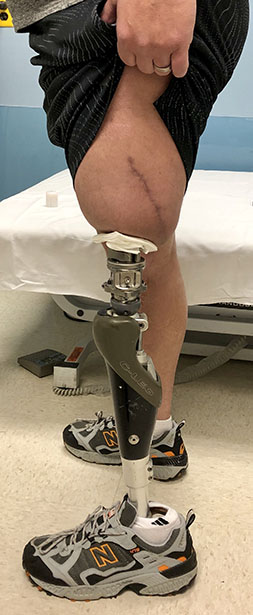
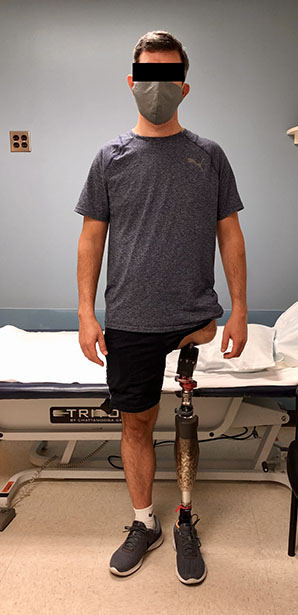
Still, many patients may undergo repeat surgical management without significant improvement with regard to prosthetic complaints. For these individuals, osseointegration (OI), which is the direct implantation of a device into the skeleton that comes through the skin and connects to the prosthetic, has been a transformative solution (Figures 1 and 2). In the 36 years since Rickard Brånemark, MD, PhD, performed the first successful transfemoral OI surgery, transcutaneous OI technology has blossomed worldwide to include cases across all forms of amputation, from thumbs to lower extremities (See Tables 1 and 2 for suggested inclusion and exclusion considerations and pros and cons for osseointegration surgery.) International thought leaders in this field have begun convening regularly to advance technologies and techniques, thanks to meetings such as the Global Collaborative Congress on Osseointegration (GCCO), among others. This orthopaedic niche continues to mature as international debate leads to improved implants, surgical techniques, and prosthetic technology. The development of OI care has encouraged the development of a peripheral human-machine interface like never before (Figure 6).
On the horizon: A new generation of prosthetics and the human-machine interface
Recent advancements have seen massive strides in how doctors connect OI patients to their surrounding environment, addressing the unique biomechanics of a direct skeletal prosthetic attachment. The integration of advanced electronics into prosthetic solutions has thus transformed how doctors address this issue. Of note — the frontier of prosthetic control has shifted toward direct myoelectric/neurologic input that commands a prosthetic. Many investigational systems attempt to implant electrodes directly into muscles and nerves of residual limbs; leads can be routed through the OI canal (in certain devices), percutaneously through the skin, or via Bluetooth to an external prosthesis. This moves beyond the limitations of surface electromyography (EMG), which can often be disrupted by sweat or soft tissue hypermobility. Implantable electrodes also allow for critical, bidirectional communication. The same sensors controlled by the nervous system can stimulate afferent nerves, allowing patients to “feel” the ground throughout the gait via stimulated sensory feedback.
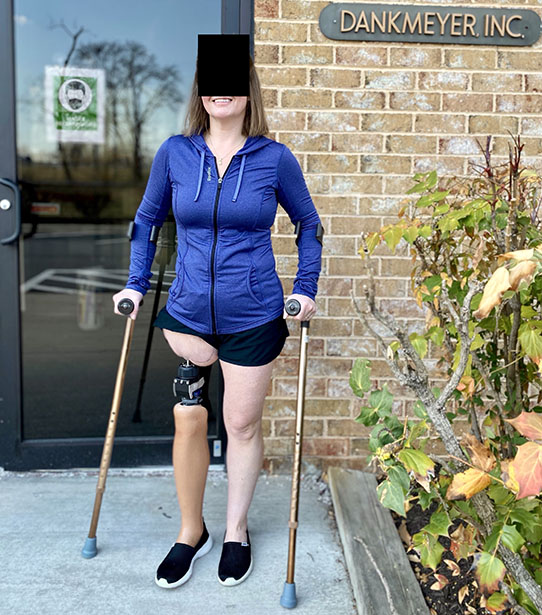
As software technology continues to evolve, patients now possess more comprehensive prosthetic control than ever. Thanks to Bluetooth technology, modern prosthetic microprocessors can be adjusted via user-controlled smartphone applications. Programmable prosthetic “modes” adapt limb function depending on the task at hand, such as walking, prolonged standing, or cycling. Such digital integration now allows amputees to seamlessly adapt their bionic limbs to specific contextual demands as needed (Figures 4 and 5).
Prosthetic companies have been able to improve these technologies thanks to direct connection, which encourages more complicated prosthetics, as weight is less of an issue than in historical sockets. The OI implant also transmits ground reaction forces directly to the femur, allowing “osseoperception” (proprioceptive feedback) to support gait. Direct skeletal loading also ameliorates osteopenia at the affected limb; strengthening the bone substrate encourages the use of more robust prosthetic machinery. As this interface between man and machine continues to refine itself, one can see a horizon that allows for “fully integrated” solutions between the biological and mechanical worlds.
Amputation care is a team sport
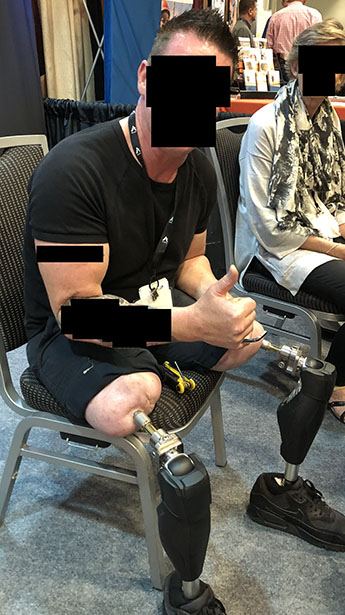
A multidisciplinary approach is the key to success for this complex amputee care. Many times, in surgery, it is most difficult to not offer a new technology or approach to a patient. OI is no different; patient selection remains the greatest tool available to providers. Collaborative clinics, which include all clinical stakeholders in amputee care, can expand the variety of solutions provided to a patient. Team members include orthopaedic surgeons, plastic surgeons, physical medicine and rehabilitation (PM&R) physicians, physical/occupational therapists, prosthetists, and (with luck) an anaplastologist. For context, anaplastologists create custom prosthetics that restore function while achieving the highest possible level of realism (Figure 3); although they traditionally focus on hyper-realistic craniofacial prosthetics, their skills can readily help in the OI clinic for patients with prosthetic cosmesis concerns. Such comprehensive teams are critical to address expectations across the continuum of care. Patients with uncontrolled hyperglycemia, smoking status, or other factors limiting bony/vascular health are not appropriate patients for such a delicate operation. OI should always be discussed on a case-by-case basis, prioritizing medical optimization and avoiding high-risk medical comorbidities whenever possible.
Conclusions: A promising future
In short, new technologies have transformed osseointegration for patients with amputations. In many ways, it can be functionally superior to socket-based prosthetics. Yet osseointegration technology must be performed, refined, and studied much more extensively before it becomes a clear first choice for most amputees. The success of modern OI, at the very least, has drastically changed the trajectory of amputee care, and more recent advancements promise a very bright future for individuals looking to achieve their lifestyle goals following amputation.
Malcolm Hamilton-Hall is a dual-degree MD/MPH student from the University of Connecticut and will graduate in May 2026. He is currently completing an orthopaedic oncology research fellowship at Johns Hopkins University. He competed in the AAOS 2026 Annual Meeting OrthoPitch finals as part of ReKinesis, a startup that has invented a gait analysis wearable device to calculate leg kinematics within two degrees of traditional video analysis.
Mark Hopkins, PT, CPO, MBA, is the CEO of Dankmeyer, Inc., the largest independent provider of orthoses and prostheses in Maryland. He is an adjunct professor at both the University of Maryland School of Medicine’s School of Physical Therapy and the Johns Hopkins University School of Medicine.
Brock Lindsey, MD, is an orthopaedic surgeon and the director of orthopaedic oncology at the Johns Hopkins Hospital. His current research focuses include osseointegration and precision immunotherapy; he is the founder of ICaPath (/EYE-SEE-A-PATH/) Incorporated.
References
- Bishay J, Yeap I, Wang T. The effectiveness of targeted muscle reinnervation in reducing pain and improving quality of life for patients following lower limb amputation. J Plast Reconstr Aesthet Surg. 2024;92:288‑298. doi:10.1016/j.bjps.2024.03.013
- Song H, Hsieh T, Yeon SH, Shu T, Nawrot M, Landis CF, Friedman GN, Israel EA, Gutierrez-Arango S, Carty MJ, Freed LE, Herr HM. Continuous neural control of a bionic limb restores biomimetic gait after amputation. Nature Medicine. 2024;30(7):2010‑2019. doi:10.1038/s41591-024-02994-9
- Karczewski AM, Zeng W, Stratchko LM, Bachus KN, Poore SO, Dingle AM. Clinical basis for creating an osseointegrated neural interface. Front Neurosci. 2022;16:828593. doi:10.3389/fnins.2022.828593
- Potter B, Rivera J, Anderson A, Souza J, Forsberg J. What functional outcomes can be expected with osseointegrated prostheses in transfemoral amputations? Clin Orthop Relat Res. 2025;483(3):513‑523. doi:10.1097/CORR.0000000000003267